Dr Wei You, Instituted of Chemistry, Chinese Academy of Sciences
Email: weiyou@iccas.ac.cn
Address: Zhongguancun North First Street 2, Beijing, China, 100190Zip: 100190
Research Areas
Solid polyelectrolytes are one of the most important energy materials interconverting chemical energy and electricity. They are essential components in a variety of energy converting devices, including fuel cell, electrolysis, electrodialysis, ion battery, redox flow battery, supercapacitor, etc. These polyelectrolytes do not conduct electrons; instead, they specifically transport cations or anions throughout the solid membrane materials. Additionally, the use of polyelectrolytes in electrochemical devices can also simplify assembly issues, avoid liquid leaking, reduce overall cost, and improve safety performance.
However, the above-mentioned electrochemical devices require different types of polyelectrolyte membranes. Thus we need to adjust the structures and functions of these membranes according to their specific requirements. The focus of our research group is to perform interdisciplinary research studying the frontiers of organic chemistry, polymer chemistry, and electrochemistry. We are using organic synthesis as a tool to prepare novel polymer materials, so that they can be used in clean and efficient energy conversion. In the Institute of Chemistry, Chinese Academy of Sciences, we are taking advantage the research platform and achievements of the Key Laboratory of Engineering Plastics to combine polymer chemistry with organic chemistry and electrochemistry.
Education
Indiana University, Bloomington, Indiana Aug 2011-May 2016
Ph.D., Chemistry
Emphasis on organic chemistry and organometallic chemistry
Tsinghua University, Beijing, China Aug 2007-July 2011
B.S., Chemistry
Emphasis on chemical biology and organometallic chemistry
Experience
Principal Investigator, Institute of Chemistry, Chinese Academy of Sciences Nov 2019-present
• Design and synthesis of polyelectrolytes with novel structures and enhanced proper
• Develop new polymerization strategies towards functional materials
Postdoctoral Associate Research, Cornell University June 2016-Oct 2019
Advisor: Prof. Geoffrey W. Coates
• Synthesized anion exchange membranes for alkaline fuel cells
• Investigated the applications of ring-opening-metathesis-polymerization (ROMP)
Publications
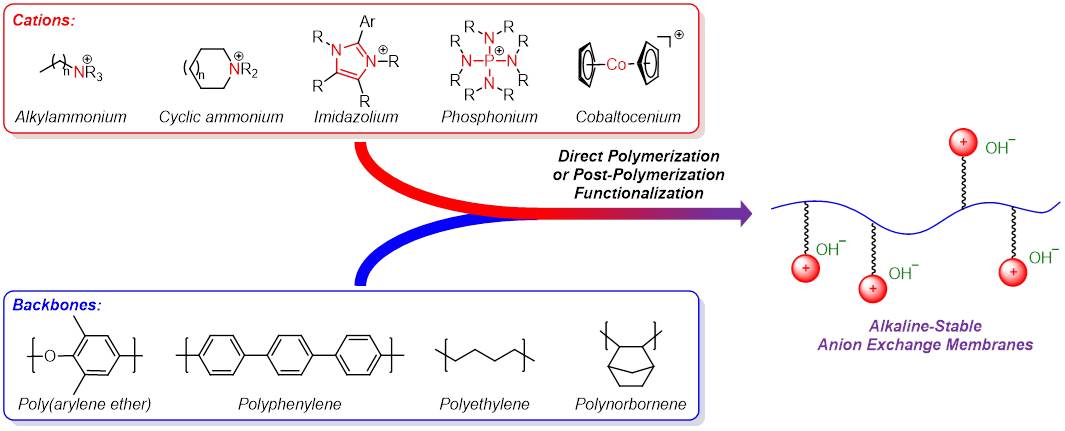
(13) You, W.; Noonan, K. J. T.;* Coates, G. W.* Alkaline-Stable Anion Exchange Membranes: A Review of Synthetic Approaches. Prog. Polym. Sci. 2020, 100, 101177.
• Invited review
(12) Hugar, K. M.;# You, W.;# Coates, G. W.* Protocol for the Quantitative Assessment of Organic Cation Stability for Polymer Electrolytes. ACS Energy Lett. 2019, 4, 1681.
# These authors contributed equally to the completion of this work.
• Invited Viewpoint.
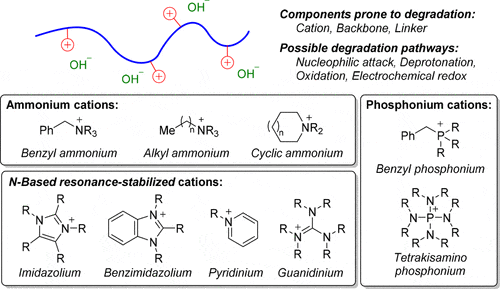
(11) You, W.; Padgett, E. S.; MacMillan, S. N.; Muller, D. A.; Coates, G. W.* Highly Conductive and Chemically Stable Alkaline Anion Exchange Membranes via ROMP of trans-Cyclooctene Derivatives. Proc. Natl. Acad. Sci.2019, 116, 9729.
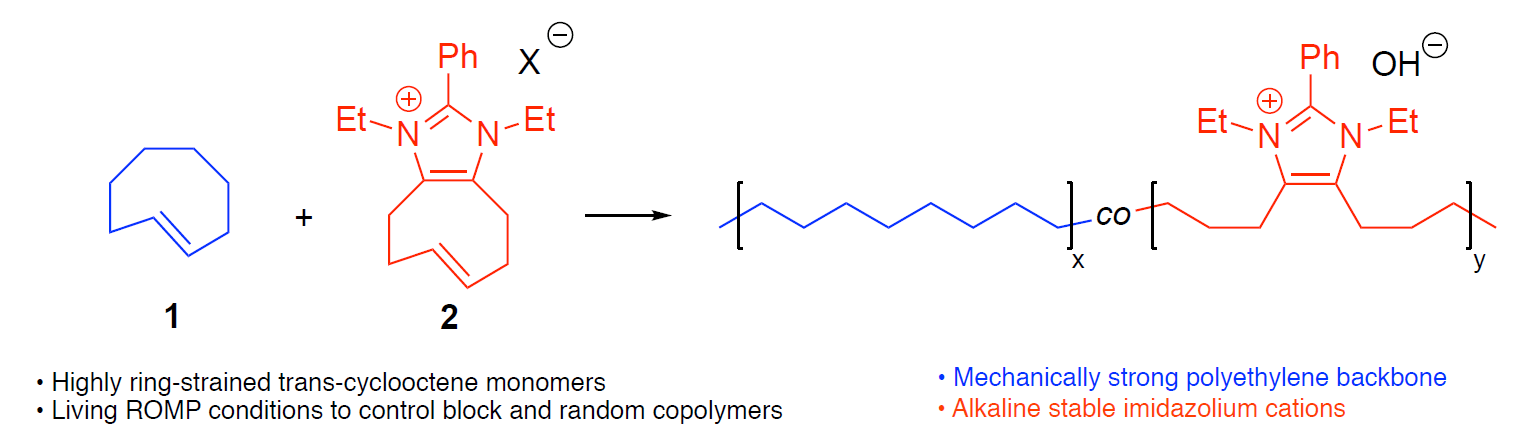
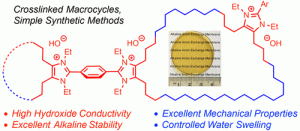
(10) You, W.; Hugar, K. M.; Coates, G. W.* Synthesis of Alkaline Anion Exchange Membranes with Chemically Stable Imidazolium Cations: Unexpected Cross-Linked Macrocycles from Ring-Fused ROMP Monomers. Macromolecules 2018, 51, 3212.
(9) Bergmann, A. M.; Oldham, A.; You, W.; Brown, M. K.* Copper-catalyzed cross-coupling of aryl-, primary alkyl-, and secondary alkylboranes with heteroaryl bromides. Chem. Commun. 2018, 54, 5381.
(8) You, W.; Brown, M. K.* Catalytic Enantioselective Diarylation of Alkenes. J. Am. Chem. Soc. 2015, 137, 14578.
• Highlighted in Synfacts: Synfacts 2016, 12, 158.

(7) You, W.; Brown, M. K.* Diarylation of Alkenes by a Cu-Catalyzed Migratory Insertion/Cross-Coupling Cascade. J. Am. Chem. Soc. 2014, 136, 14730.

(6) Smith, K. B.; Logan, K. M.; You, W.; Brown, M. K.* Carboboration of Alkenes Enabled by Synergistic Catalysis. Chem. Eur. J. 2014, 20, 12032.
(5) Zhou, Y.-Q.;# You, W.;# Smith, K. B.; Brown, M. K.* Copper-Catalyzed Cross-Coupling of Boronic Esters with Aryl Iodides and Application to Carboboration of Alkynes. Angew. Chem., Int. Ed. 2014, 53, 3475.
# These authors contributed equally to the completion of this work.
• Highlighted in Synfacts: Synfacts 2014, 10, 625.

(4) You, W.; Li, Y.; Brown, M. K.* Stereoselective Synthesis of All-Carbon Tetrasubstituted Alkenes from In Situ Generated Ketenes and Organometallic Reagents. Org. Lett. 2013, 15, 1610.
• Highlighted in Synfacts "Synfact of the month": Synfacts 2013, 9, 765.
• Highlighted in ChemInform2013,44, DOI: 10.1002/chin.201332056.

(3) Liao, Q.; You, W.; Lou, Z.-B.; Wen, L.-R.; Xi, C.* Copper-Catalyzed Tandem S-Alkylation and S-Alkenylation of Sodium Sulfide: Synthesis of 2,3-Dihydrothiophenes and Thiophenes.Tetrahedron Lett. 2013, 54, 1475.
(2) You, W.; Yan X.; Liao, Q.; Xi, C.* Cu-Catalyzed Double S-Alkenylation of Potassium Sulfide: A Highly Efficient Method for the Synthesis of Various Thiophenes. Org. Lett. 2010, 12, 3930.
• Highlighted in ChemInform2010,41, DOI: 10.1002/chin.201052107.

(1) Xi, C.;* Yan, X.; You, W.; Takahashi, T.* Coupling Reactions of Zirconate Complexes Induced by Carbonyl Compounds. Angew. Chem., Int. Ed. 2009, 48, 8120.
• Highlighted in ChemInform2010,41, DOI: 10.1002/chin.201007049.